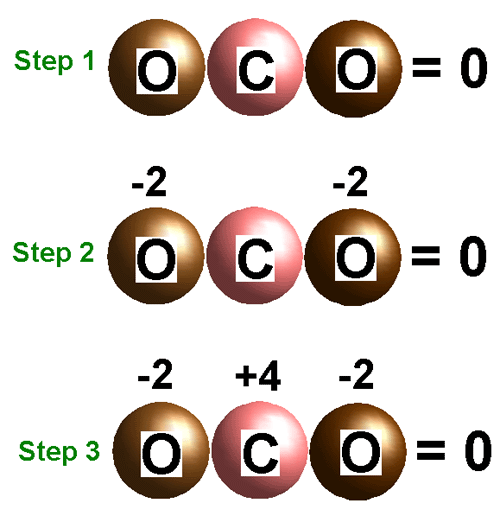Chapter 8 Vocab
Alloy: A mixture of elements that has metallic properties.
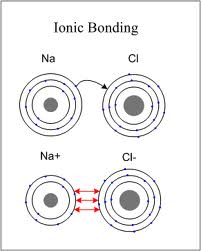
Anion: Another name for a negatively charged ion.
Cation: Another name for a positively charged ion.
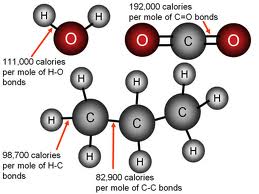
Chemical Bond: The force that holds two atoms together.
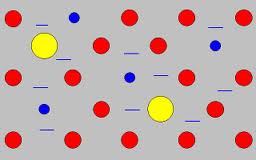
Delocalized Electrons: Atoms that are free to move.
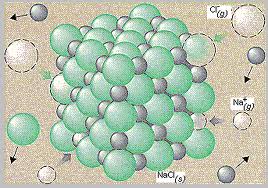
Electrolyte: An ionic compound whose aqueous solution, conducts and electric movement.
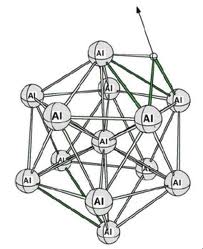
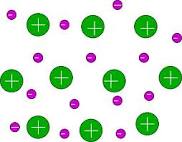
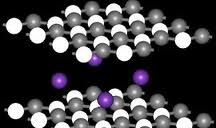
Electron Sea Model: States that all metal atoms in a metallic solid contribute their valence electrons to form a sea of electrons.
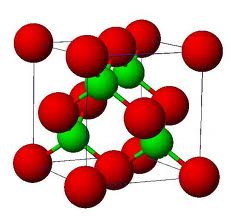
Formula Unit: The easiest ratios of the ions represented in an ionic compound.
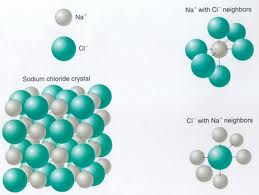
Lattice Energy: The energy that is required to separate one mole of the ions of an ionic compound.
Monatomic Ion: A one atom ion that indicates the charges of the atoms due to the location on the periodic table.
Oxidation Number: The charge of the Monatomic Ions.
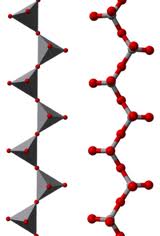
Oxyanion: A polyatomic ion composed of an element, bonded to one or more oxygen atoms.
Polyatomic Ion: Ions made up of more than one atom.
Alloy: A mixture of elements that has metallic properties.
Anion: Another name for a negatively charged ion.
Cation: Another name for a positively charged ion.
Chemical Bond: The force that holds two atoms together.
Delocalized Electrons: Atoms that are free to move.
Electrolyte: An ionic compound whose aqueous solution, conducts and electric movement.
Electron Sea Model: States that all metal atoms in a metallic solid contribute their valence electrons to form a sea of electrons.
Formula Unit: The easiest ratios of the ions represented in an ionic compound.
Lattice Energy: The energy that is required to separate one mole of the ions of an ionic compound.
Monatomic Ion: A one atom ion that indicates the charges of the atoms due to the location on the periodic table.
Oxidation Number: The charge of the Monatomic Ions.
Oxyanion: A polyatomic ion composed of an element, bonded to one or more oxygen atoms.
Polyatomic Ion: Ions made up of more than one atom.